Lifespan Rhythms of Memory and Cognition (RHYME)
Research Scientists
Markus Werkle-Bergner
Elisa Buchberger
Martin J. Dahl (until 11/2022)
Ann-Kathrin Joechner
Ulman Lindenberger
Beate Mühlroth (until 04/2020)
Chi (Zoe) Ngo
Sarah Power (as of 04/2021)
Early Childhood Memories (ECHO)
Charting the Ontogeny of Memory Component Processes Across Early Childhood (COMIC)
Child and Adolescent Age Differences in Rhythmic Brain Activity During Sleep
The RHYME project investigates lifespan differences and changes in attention, working memory, and episodic memory at structural, functional, and behavioral levels of analysis. It combines experimental with longitudinal research designs and uses multimodal data from a wide range of neuroimaging methods. The project collaborates closely with the Max Planck Partner Group Hippocampal Circuit and Code for Cognition Lab (HCCCL), led by former postdoc Attila Keresztes, which was established in 2021 at the Brain Imaging Centre of the Research Centre for Natural Sciences in Budapest, Hungary. During the reporting period, the project’s primary research focus was on the design and administration of two series of studies, Early Childhood Memories (ECHO) and Charting the Ontogeny of Memory Component Processes Across Early Childhood (COMIC). Informed by animal and computational models of memory, ECHO and COMIC serve to elucidate the ontogeny of episodic memory in infancy and middle childhood, respectively. Both series of studies represent long-term efforts that will take several years to come to full fruition. A second major portion of the project’s research activities, focusing on noradrenergic neuromodulation and the locus coeruleus, has become part of the LINE project. RHYME also continued its earlier work on the development of rhythmic brain activity during childhood and adolescence.
Early Childhood Memories (ECHO)
Human infants show clear learning from an early age, but forget episodic memories formed in the first 2 to 3 years of life. This phenomenon, called infantile amnesia, appears to be evolutionarily preserved, as it can be found across a wide range of species. However, its function and mechanisms are only poorly understood. In non-human animals, memory engrams can be probed directly with invasive neuroscience technologies that cannot be used in human research. To bridge the ensuing gap in knowledge, we have initiated a collaboration with Tomás Ryan (Trinity College Dublin). The goal of the collaboration is to build a coordinated research program on the ontogeny of memory engrams in rodents and humans. In particular, we hope to identify non-invasive behavioral and neural markers of memory engram ensemble activity that are applicable to human infants and children. If successful, the availability of such markers will allow us to develop and evaluate interventions that re-activate or suppress specific memories, including those formed during the first year of life.
The research is guided by three main objectives: First, we have developed object–context and maze-learning assays to study infantile amnesia in rodents that can inform and parallel human behavioral experiments. Second, we aim to identify biomarkers of memory expression in rodent infants that can be translated to human infants. Here, we focus on (mobile) EEG as a bridge marker of neural activity in both animal models and human infants. Third, our long-term goal comprises the validation of non-invasive interventions for the reinstatement of infant memory engrams in humans.
For the human studies, we built a dedicated testing site (see Figure 1). In a large room, we constructed a testing arena, about 4 m x 4 m in size, enclosed by walls. The arena features several hiding spots that indicate the different spatial locations the children should learn. The walls are constructed as projection screens to dynamically create unique “natural” environments (e.g., a playground, a beach) across different learning trials. Using this setup, we provide infants with a unique learning experience shortly before the offset of infantile amnesia (at 18–24 months of age). The successful formation of a unique memory trace is assured by a retest one day later. The natural effects of forgetting are assessed 30 to 60 days later. The task infants perform is modeled according to a context-location memory task typically used for mice (i.e., a Barnes maze).

Charting the Ontogeny of Memory Component Processes Across Early Childhood (COMIC)
In a changing environment, an adaptive memory system needs to gauge the conflicting demands of extracting commonalities across experiences to generate novel inferences (i.e., generalization) while simultaneously forming discriminable representations of specific events (i.e., memory specificity). Computational memory models suggest that specific experiences are initially encoded as hippocampus-dependent episodic memories and slowly become amenable to generalization through consolidation. This process is typically facilitated by post-learning sleep. Early in life, children are poor at remembering the details of specific events, yet display a remarkable capacity to extract schematic knowledge about the environment. Such observations suggest a developmental lead–lag relation between generalization and memory specificity. In two ongoing large-scale studies with more than 500 4- to 8-year-old children, we aim to delineate the ontogeny of memory specificity and generalization across development in early middle childhood.
The first study tests the conjecture that the uneven maturation of intra-hippocampal structures is linked longitudinally to the late emergence of memory specificity in childhood. Here, we also ask to what extent sleep-dependent memory consolidation amplifies the reliance on fast generalization in early childhood at the expense of memory specificity. To this end, we have implemented an accelerated longitudinal design with 200 children aged 4, 5, or 6 years old at the first measurement occasion. At each of the three annual measurement time points, participants will take part in up to five sessions that also include structural MRI measurements. We have adapted a novel memory task (the Collection Game Task) that allows developmentally adequate assessments of generalization, context-binding, and contextual as well as perceptual specificity. An initial cross-sectional validation study supports the hypothesis that age differences are more pronounced for generalization than for memory specificity during early childhood (see Figure 2).
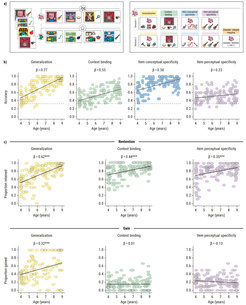
Figure 2. Assessing component memory processes in middle childhood. (a) Schematic illustration of the Collection Game Task used to assess generalization and context binding, as well as conceptual and perceptual specificity longitudinally in 4- to 8-year-old children. During encoding, the children watch a stream of visual images consisting of a background scene, a central character, and an associated object. Children are told that each of the characters goes to different places and collects the things they want for their collection. Hence, each character (e.g., Luntik) is shown on a specific kind of background scene (e.g., stages), and paired with a specific class of objects (e.g., musical instruments). Throughout the encoding phase, each character appears in four trials in an intermixed order, each time with a different context and object. During the test, four tasks are conducted. To assess generalization, a character is shown with three objects, of which only one is appropriate for their collection (e.g., a not-yet-seen musical instrument). To measure context binding, the same character–scene pairing as experienced during encoding is shown again. The children’s task is to identify the exact object that was shown along with this character–scene combination. Conceptual and perceptual specificity are probed by identifying the previously encountered object among similar objects along with its precise perceptual features. All images in (a) from flaticon.com. (b) Results of the Collection Game Task on the immediate test of the first day. All facets of memory performance were positively correlated with age. As hypothesized, the age association was stronger for generalization than for memory specificity. (c) Delayed performance after one night of sleep. Items that resulted in a correct response in the evening and were also answered correctly in the morning were classified as maintained items. Items that did not yield a correct response in the evening but were correctly answered the next morning were classified as gained. With increasing age, children were more likely to maintain both generalized and specific memory contents. For generalized memories, overnight gains increased with age, while gains for specific mnemonic details were weak across the entire age range.
Image: Elisa Buchberger & Ann-Kathrin Joechner / MPI for Human Development
Adapted from Buchberger, Joechner et al. (2022)
Original image licensed under CC BY 4.0
Neurocomputational models postulate that memory performance reflects the interplay of generalization, pattern separation, and pattern completion. However, it is unknown whether this tripartite structure is already fully established in early childhood. Based on an in-depth review of the literature on memory development during childhood, we assembled and designed a child-adequate battery of seven tasks yielding 13 indicators, with the aim of establishing latent factors of generalization, pattern separation, and pattern completion. The experimental procedures and analysis strategy of the study have been accepted as a Stage 1 Registered Report by Developmental Science. The overall goal of the study is to formally test whether the postulated tripartite process structure of memory is already present in 4- to 6-year-old children.
Key Reference
Child and Adolescent Age Differences in Rhythmic Brain Activity During Sleep
Following up on our earlier work on memory consolidation during sleep, we investigated the temporal coordination between slow oscillations and spindles in children and adolescents during sleep. In young adults, coupling of rhythmic brain activity during sleep is most pronounced for fast spindles (≈ 12.5–16 Hz), while slow spindles (≈ 9–12.5 Hz) synchronize shifted in time. Interestingly, slow sleep spindles, rather than fast sleep spindles, prevail in childhood and early adolescence. While both sleep spindles and slow oscillations are known to show developmental changes, the ontogeny of their co-occurrence has remained elusive. In a collaboration with Kerstin Hoedlmoser, Paris Lodron University Salzburg, we investigated age differences in slow oscillation–spindle coupling, the key electrophysiological mechanism supporting memory consolidation in young adults, in three samples of children and adolescents (Joechner et al., 2022). Corroborating previous findings, we identified fast sleep spindles in all age groups. However, specifically in young children, the dominant type of fast spindles was nested with the slow spindle range, and only few adult-like fast spindles were detectable. In older children and adolescents, fast spindles were more numerous and more closely resembled adult-like fast spindles. Astonishingly, temporal couplings to slow waves were barely discernable for ontogenetically early fast spindles, while activity in the adult-like fast spindles range was clearly synchronized with the up-peak of slow oscillations. These results suggest that the increasing ability to generate adult-like fast sleep spindles facilitates the emergence of slow oscillation-spindle couplings during childhood and adolescence (see Figure 3).
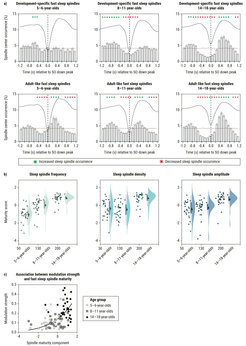
Figure 3.The emerging coordination between slow oscillations and spindles during sleep in childhood. (a) Peri-event time histograms capturing the co-occurrence between development-specific (upper row) and adult-like fast spindles (lower row) with slow oscillations. Gray bars indicate the percentage of spindle occurrence in a given time-bin relative to the down-peak of the corresponding slow oscillations. Compared to a randomly shuffled surrogate distribution, there is no indication of increased co-occurrence of development-specific spindles with the slow oscillation up-peak in younger children. However, a clear modulation pattern can be observed for adult-like fast spindles. (b) With increasing age, development-specific fast spindles increasingly resemble the fast spindles typically observed in adults. Accordingly, differences between individual development-specific spindles and those identified in the adult-like fast spindle range decrease in frequency, density, and amplitude with age. (c) The more individually identified fast spindles in children resemble adult-like fast spindles (as captured in an increasing multivariate maturity score, see Joechner et al., 2022), the more pronounced the synchronization pattern between spindles and slow oscillations (expressed via the Kullback-Leibler divergence between the actual peri-event time histogram distributions and a randomly shuffled surrogate distribution).
Image: MPI for Human Development
Adapted from Joechner et al. (2022)
Original image licensed under CC BY-NC-ND 4.0
Key References


