Brain Imaging Methods in Lifespan Psychology
(Concluding Report)
Research Scientists
Nils Bodammer
Davide Santoro
Ulman Lindenberger
Naftali Raz
Research on human development seeks to delineate the variable and invariant properties of age-graded changes in the organization of brain–behavior–environment systems. Magnetic resonance imaging (MRI) modalities, including magnetic resonance spectroscopy (MRS), are an indispensable part of this effort, as they allow for the noninvasive assessment of brain function, anatomy, microstructure, and metabolism.
The goals of the Brain Imaging Methods project, which was founded in 2011, have been twofold: to ascertain and improve the measurement quality of standard brain imaging protocols at the Center, and to complement the standard imaging repertoire by advanced sequences with enhanced interpretability that hold promise in elucidating structural changes and physiological mechanisms related to maturation, learning, and senescence. In pursuing these goals, the project has served as a resource for other projects interested in imaging (e.g., Porat et al., 2022; Wenger et al., 2022). Hence, structural and quantitative MRI methods occupy a central place in the project.
During the reporting period, the project has focused on: (a) T1 mapping by means of an MP2RAGE acquisition together with a B1 map-correction protocol to obtain estimates of laminar myelination across the cortical sheet; (b) myelin water fraction (MWF) imaging; (c) multiband-accelerated high-angular-resolved diffusion imaging (HARDI); and (d) neuromelanin-sensitive high-resolution imaging of the brainstem to determine the individual position and extension of the loci coerulei (Dahl, Bachman, et al., 2023).
With the establishment of the Institute’s MR Physics Research Group and Core Facility in early 2023, the brain-imaging project within the Center for Lifespan Psychology has come to an end. Nils Bodammer and Davide Santoro have joined the Core Facility. The establishment of the Core Facility and the installation of a new 3-Tesla and a 7-Tesla scanner in the coming 2 to 3 years will further strengthen the brain-imaging expertise at the Institute.
T1 Mapping Using MP2RAGE With B1 Map Correction
The longitudinal relaxation time T1 in cortex is affected by the myelin content of the laminae. T1 mapping offers a noninvasive method to determine cortical structures and their changes over time. Our original protocol for the accurate estimation of T1 maps in the brain made use of three repeated MP2RAGE sequences developed by José Marques and colleagues, with tailored paired values for the inversion times to cover the whole range of T1 values in the brain. However, this original protocol was rather time-consuming and prone to motion artefacts. A close look at the MP2RAGE signal evolution function allowed us to optimize the protocol by specifying one single acquisition with maximal sensitivity in the region of the white matter versus gray matter contrast. Within only 10 minutes acquisition time at our 3-Tesla scanner, we have been able to reach submillimeter resolution (0.8 mm isotropic) showing exquisite details of the cortical thickness structure. The resulting T1 map is then corrected by a B1 map to cancel hardware imperfections and radio frequency inhomogeneities across the brain (see Figure 1). The B1 mapping method used, based on earlier work by Davide Santoro and others, was developed in-house, and its duration was reduced to only 10 seconds. The in-house B1 mapping method has been shared with other MRI centers (such as Wayne State University, Detroit, USA), and might give rise to possible future collaborations, especially with MRI centers equipped with a 7-Tesla scanner, where the B1 map correction is essential.
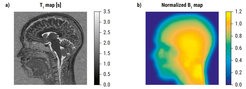
Figure 1. (a) Typical T1 map of a young adult’s brain (One slice out of a 3D volume with matrix 270 x 300 x 200) obtained using MP2RAGE and B1 map correction. Total acquisition time was 10 minutes and 20 seconds. (b) Normalized B1 map used for correction, resized and co-registered with the T1 map. Original matrix 56 x 60 x 18, Acquisition time 15 seconds.
Myelin-Water Fraction Imaging (MWF)
Based on a time series of T2-weighted MR images with increasing echo-times, MWF imaging evaluates the transversal relaxation in a multiexponential manner. The fraction of short T2s (< 40 ms) provides an estimate of the portion of water molecules located between myelin sheaths, presumably reflecting the degree of myelination within white matter. Work on MWF imaging has been done in collaboration with Jeffrey A. Stanley (Wayne State University, Detroit, USA).
High Angular Resolved Diffusion Imaging (HARDI)
Diffusion imaging captures the movement of water molecules, termed diffusion. Diffusion in tissue is hindered by cell membranes. Therefore, the orientation-dependent diffusion profiles provide information about tissue microstructure, namely about the density of neural fibers for any voxel in brain tissue as a function of their orientation. Special MR protocols sensitized to the diffusion of water molecules in tissue allow measurement of such diffusion orientation profiles. The optimization of either spatial or angular resolutions of such acquisition protocols has been performed for multiband-accelerated diffusion imaging protocols at 3 Tesla in collaboration with Claudia Wolf (Freie Universität Berlin).
